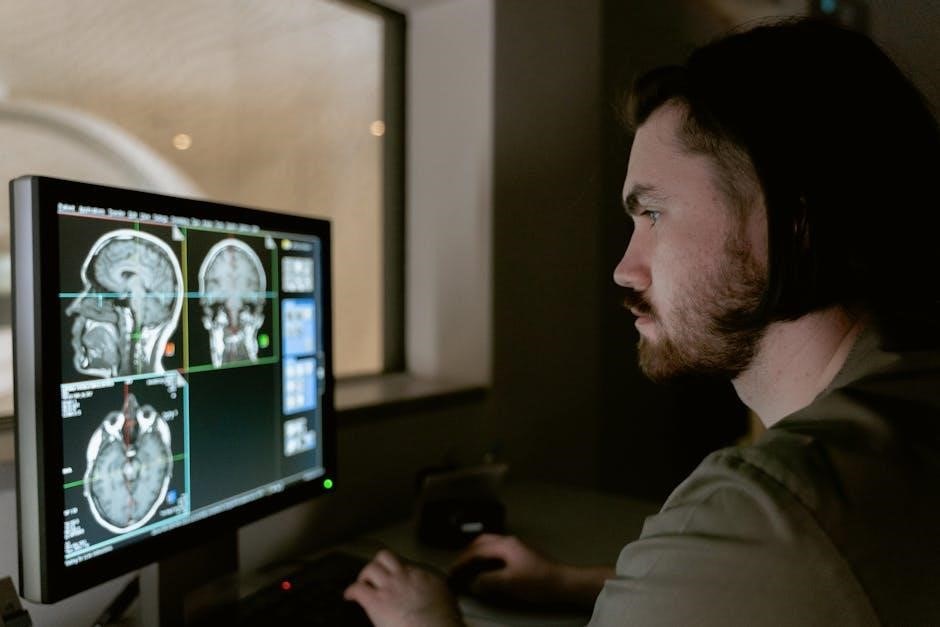
LINQ II MRI Technical Manual: Comprehensive Overview
This manual details MRI compatibility for the LINQ II Insertable Cardiac Monitor (ICM) models M301, M302, and M312, ensuring patient safety during scans.
The LINQ II Insertable Cardiac Monitor (ICM), manufactured by Medtronic, represents a significant advancement in long-term cardiac rhythm monitoring. This small, implantable device continuously records the heart’s electrical activity, providing valuable diagnostic information for detecting infrequent arrhythmias. Crucially, the LINQ II boasts a battery life of up to 4.5 years, minimizing the need for premature device replacements.
A key feature distinguishing the LINQ II is its MRI Conditional designation, allowing patients to undergo magnetic resonance imaging (MRI) scans when medically necessary. However, strict adherence to the guidelines outlined in this technical manual is paramount to ensure patient safety and maintain device functionality. The device models M301, M302, and M312 are specifically addressed within these guidelines.
Purpose of the MRI Technical Manual
This LINQ II MRI Technical Manual serves as a comprehensive resource for healthcare professionals involved in the care of patients with an implanted LINQ II Insertable Cardiac Monitor who require Magnetic Resonance Imaging (MRI). Its primary objective is to provide detailed instructions and safety guidelines to mitigate potential risks associated with MRI scans.
The manual outlines specific parameters, protocols, and precautions necessary to ensure both patient safety and the continued optimal performance of the LINQ II device. It addresses potential artifacts that may arise during scanning, and strategies to minimize their impact on image quality and clinical interpretation. Adherence to this manual is crucial for maintaining FDA clearance and compliance with MRI safety standards.

MRI Compatibility: Core Principles
LINQ II ICMs (M301, M302, M312) are MR Conditional, requiring adherence to specific conditions of use to ensure patient safety during MRI procedures.
Understanding MR Conditional Designation
The “MR Conditional” designation for the LINQ II Insertable Cardiac Monitor (ICM) signifies that the device is safe for use in a Magnetic Resonance (MR) environment when specific conditions are met. This is not the same as “MR Safe.” MR Conditional means the device has been tested and demonstrated to be non-detrimental under defined scanning parameters.
Crucially, strict adherence to the guidelines outlined in the LINQ II MRI Technical Manual is paramount. These conditions encompass magnetic field strength limitations, Specific Absorption Rate (SAR) limits, and scan sequence considerations. Failure to comply could result in device malfunction, including electrical reset or inappropriate sensing.
Regularly reviewing updated information regarding MR Conditional status and scan parameters is essential, as these can evolve with new data and regulatory updates.
Specific LINQ II Models and MRI Compatibility (M301, M302, M312)
The LINQ II Insertable Cardiac Monitor (ICM) models M301, M302, and M312 are all designated as MR Conditional, allowing patients to undergo Magnetic Resonance Imaging (MRI) scans under strictly defined conditions. This compatibility is a significant advantage, as it avoids the need for device explantation prior to necessary imaging.
However, it’s vital to confirm the specific model number implanted in the patient before proceeding with an MRI. The MRI Technical Manual details the precise parameters applicable to each model.
Verification of the model number (LUX-Dx, LUX-Dx II, or LUX-Dx II) is a critical step in the pre-scan assessment process, ensuring appropriate safety protocols are followed.
Battery Life Considerations & MRI Scans (Up to 4.5 years)
The LINQ II Insertable Cardiac Monitor boasts an impressive battery life of up to 4.5 years, providing a substantial window for monitoring and diagnostic capabilities. This extended longevity minimizes the frequency of device replacements, enhancing patient convenience and reducing intervention needs.
Importantly, the device’s battery status does not directly impact its MRI compatibility. Patients can safely undergo MRI scans throughout the device’s operational lifespan, provided all specified conditions outlined in the MRI Technical Manual are meticulously followed.
However, it’s crucial to document the battery status at the time of the MRI scan as part of the comprehensive scan record.

Pre-MRI Scan Assessment
Prior to any MRI scan, thorough patient eligibility verification, device model confirmation (M301, M302, M312), and MRI environment specification review are essential.
Patient Eligibility Verification
Confirming patient suitability for MRI is paramount. This involves a detailed review of the patient’s medical history, focusing on any contraindications to MRI procedures. Specifically, assess for the presence of non-MRI conditional implants or devices.
Verify the LINQ II device model number – M301, M302, or M312 – as MRI compatibility is contingent upon these specific models. Ensure the device was implanted according to Medtronic’s instructions for use.
Crucially, adherence to the Conditions of Use, as outlined in the LINQ II MRI Technical Manual, must be verified before each scan to ensure the most current safety information is applied.
Review of Implanted Device Model Number
Accurate identification of the implanted LINQ II model is critical for MRI safety. Only models M301, M302, and M312 are designated as MRI Conditional, and scanning a different model could pose significant risks to the patient.
This verification should be documented in the patient’s medical record, alongside the date of implantation. Confirm this information with the patient’s implant card or previous medical reports.
Failure to confirm the correct model number before proceeding with an MRI scan could lead to device malfunction, inaccurate imaging, or patient injury. Always cross-reference with the official Medtronic documentation.
Confirmation of MRI Environment Specifications
Prior to any MRI scan, meticulous verification of the MRI environment is paramount. The LINQ II ICM has specific requirements regarding magnetic field strength, Specific Absorption Rate (SAR) limits, and scan sequence parameters.
Ensure the MRI system meets these specifications, and that all personnel are aware of the LINQ II’s conditional status. Document the scanner’s field strength (Tesla) and the SAR settings used during the scan.
Deviation from these specifications could compromise device functionality and patient safety. Adherence to the LINQ II MRI Technical Manual is non-negotiable.
MRI Scan Parameters & Protocols
Strict adherence to defined parameters is crucial. This includes magnetic field strength limitations and SAR limits to ensure LINQ II device safety during MRI.
Magnetic Field Strength Limitations
The LINQ II Insertable Cardiac Monitor is designated as MR Conditional, but specific limitations regarding magnetic field strength must be strictly observed to maintain patient safety and device functionality. Scans should only be performed in MRI environments adhering to the conditions outlined in the LINQ II MRI Technical Manual.
Generally, the device is compatible with 1.5 Tesla and 3.0 Tesla MRI systems. However, exceeding these field strengths is prohibited and may lead to device malfunction or inaccurate data. Prior to any scan, confirm the MRI system’s field strength and ensure it falls within the approved range. Deviation from these guidelines could compromise the integrity of the LINQ II system and potentially endanger the patient.
Specific Absorption Rate (SAR) Limits
Maintaining adherence to Specific Absorption Rate (SAR) limits is crucial during MRI scans with patients implanted with the LINQ II Insertable Cardiac Monitor. SAR represents the rate at which radiofrequency energy is absorbed by the body. Exceeding established SAR limits can cause tissue heating and potentially harm the patient.
The LINQ II MRI Technical Manual provides detailed guidance on acceptable SAR levels based on the scan parameters and body region being imaged. Operators must configure the MRI system to operate within these specified limits. Regular monitoring of SAR values during the scan is recommended to ensure continued compliance and patient safety. Ignoring SAR limitations poses a significant risk.
Scan Sequence Considerations
Careful selection of MRI scan sequences is paramount when imaging patients with the LINQ II Insertable Cardiac Monitor. Certain sequences may induce greater radiofrequency energy deposition, potentially affecting device performance or causing localized heating. The LINQ II MRI Technical Manual outlines recommended scan sequences and those to avoid, prioritizing patient safety and data integrity.
Minimizing scan time and utilizing sequences with lower energy output are generally advised. Gradient echo sequences, for example, may be preferable to spin echo sequences in some cases. Thorough understanding of sequence parameters and their potential impact on the LINQ II device is essential for optimal imaging and risk mitigation.
Potential Artifacts & Image Quality
The LINQ II device can cause foreign body artifact on MRI and CT scans, potentially impacting clinical interpretation; however, strategies exist to minimize these effects.
Foreign Body Artifact Associated with LINQ II
The presence of the LINQ II Insertable Cardiac Monitor inevitably introduces foreign body artifact during thoracic CT and cardiac MRI scans. This artifact manifests as geometric distortions and signal voids in the immediate vicinity of the device. The degree of artifact is influenced by several factors, including the magnetic field strength utilized during MRI, the specific scan sequence employed, and the device’s position within the patient’s chest.
Generally, the artifact appears as a localized area of signal loss or distortion, potentially obscuring adjacent anatomical structures. Careful consideration must be given to this artifact when interpreting images, as it could mimic or mask underlying pathology. A tertiary center experience highlights the need for assessment of both the artifact’s degree and its clinical significance.
Impact of Artifact on Clinical Interpretation
The foreign body artifact created by the LINQ II ICM can significantly impact clinical interpretation of cardiac MRI and thoracic CT scans. This artifact’s presence may obscure critical anatomical details, potentially leading to misdiagnosis or delayed detection of pathology. Clinicians must be aware of the potential for artifact to mimic cardiac or pulmonary abnormalities, necessitating careful image evaluation.
Specifically, the artifact can interfere with accurate assessment of ventricular function, wall motion, and the presence of perfusion defects. Furthermore, it may complicate the evaluation of mediastinal structures and lung parenchyma. Thorough understanding of the artifact’s characteristics is crucial for differentiating it from true pathology and ensuring accurate diagnostic conclusions.
Strategies for Minimizing Artifact
Several strategies can be employed to minimize the foreign body artifact associated with the LINQ II ICM during MRI and CT scans. Optimizing scan parameters, such as adjusting slice thickness and employing appropriate reconstruction algorithms, can reduce artifact intensity. Utilizing techniques like saturation bands or spectral presaturation can also be beneficial in suppressing artifact.
Careful patient positioning, avoiding direct alignment of the device within the magnetic field, may further mitigate artifact. Post-processing techniques, including artifact reduction filters, can be applied to improve image quality. Clinicians should collaborate with MRI technologists to implement these strategies and achieve optimal image clarity for accurate diagnosis.
Patient Safety Considerations
Patients with LINQ II should avoid diathermy, high radiation, electrosurgical cautery, and lithotripsy to prevent device reset or inappropriate sensing during procedures.
Avoiding Diathermy, Radiation & Electrosurgical Cautery
It is crucial to protect the LINQ II Insertable Cardiac Monitor from potential interference caused by various energy sources. Specifically, patients must avoid exposure to diathermy, which utilizes high-frequency electromagnetic currents for therapeutic heating, as this can induce electrical reset within the device. Similarly, high sources of radiation, including those encountered during certain medical procedures, pose a risk.
Electrosurgical cautery, employing high-frequency electrical current to cut or coagulate tissue, also presents a hazard. These energy sources can lead to inappropriate sensing, potentially causing the device to misinterpret signals and deliver unintended therapy. Strict adherence to these precautions, as outlined in the Medical Procedure and EMI Warnings manual, is paramount for maintaining patient safety and device functionality.
Electrical Reset & Inappropriate Sensing Risks
The LINQ II Insertable Cardiac Monitor is susceptible to electrical reset and inappropriate sensing when exposed to certain electromagnetic interference (EMI). Sources like diathermy, high-intensity radiation, and electrosurgical cautery can disrupt the device’s normal operation. Electrical reset involves the device reverting to its factory settings, potentially requiring reprogramming.
Inappropriate sensing occurs when the device misinterprets signals as originating from the heart, leading to unnecessary pacing or inhibition of therapy. These risks are detailed in the Medical Procedure and EMI Warnings manual. Careful consideration and adherence to recommended precautions are vital to mitigate these potential complications and ensure continued reliable device performance for the patient.
Post-Scan Device Check
Following an MRI scan involving a patient with a LINQ II Insertable Cardiac Monitor, a thorough device interrogation is crucial. This check verifies the device’s continued functionality and confirms that no unintended programming changes or adverse effects occurred during the scan. The interrogation should assess battery status, sensing thresholds, and lead integrity.
Any observed anomalies require immediate attention and potential reprogramming. Documentation of the post-scan check, including date, time, and findings, is essential for maintaining a complete patient record. Prompt evaluation ensures optimal device performance and patient safety post-procedure, confirming the LINQ II remains reliable.
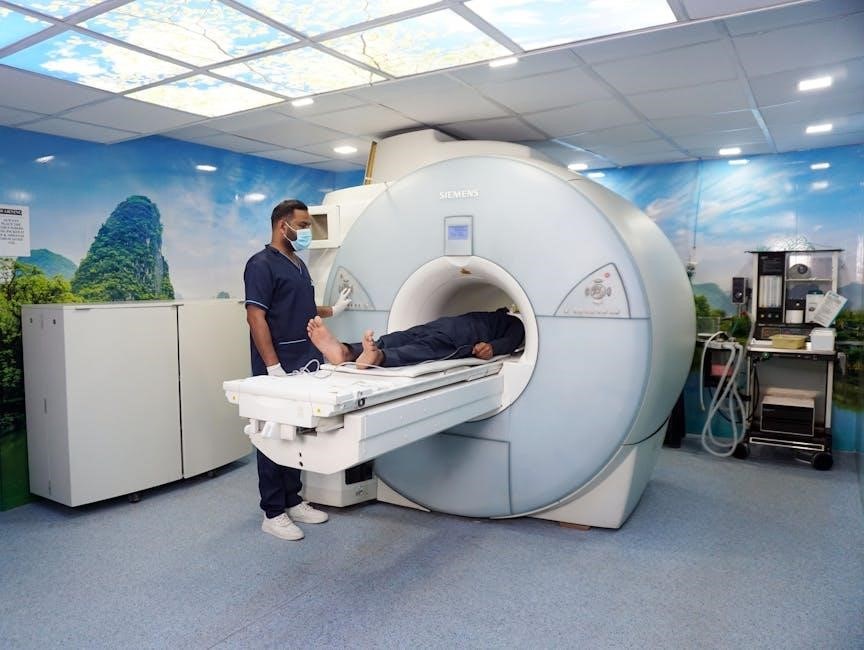
Procedure-Specific Guidelines
Specific protocols are needed for thoracic CT scans, cardiac MRI scans, lithotripsy, and therapeutic ultrasound when a LINQ II device is present.
Thoracic CT Scans with LINQ II
Thoracic CT scans can be performed in patients with implanted LINQ II devices, but careful consideration of potential artifacts is crucial. A case series review highlights the need to assess the degree of foreign body artifact created by the LINQ II during imaging. While generally not clinically significant, the artifact’s presence should be acknowledged and understood by the interpreting physician.
Image reconstruction techniques may help minimize artifact. Clinicians should prioritize patient safety and data preservation during these scans. Documentation of the LINQ II presence and any observed artifact is essential for accurate interpretation and future reference. The device’s location should be noted relative to anatomical structures.
Cardiac MRI Scans with LINQ II
Cardiac MRI scans are permissible in patients with implanted LINQ II devices, provided strict adherence to the LINQ II MRI Technical Manual’s guidelines. A tertiary center experience demonstrates that these scans can be performed safely, but careful evaluation of foreign body artifact is paramount. The degree of artifact, and its impact on clinical interpretation, must be assessed.
Prior to scanning, confirm patient eligibility and MRI environment specifications. The LINQ II allows patients to undergo MRI when needed, but adherence to conditions of use is vital. Documentation of the scan parameters and any observed artifact is crucial for accurate diagnosis and ongoing patient management.
Lithotripsy & Therapeutic Ultrasound Precautions
Patients with the LINQ II Insertable Cardiac Monitor should avoid therapeutic ultrasound and lithotripsy procedures due to the potential for device malfunction. These modalities can deliver high energy levels that may cause electrical reset or inappropriate sensing of the implanted device. The Medical Procedure and EMI Warnings, Precautions and Guidance Manual details these risks.
Careful consideration must be given to alternative treatment options. If lithotripsy or therapeutic ultrasound is unavoidable, thorough documentation of the procedure and post-procedure device interrogation are essential. Maintaining awareness of these potential interactions is critical for ensuring patient safety and optimal device performance.

Emergency Procedures
In the event of a LINQ II device malfunction during or after an MRI, immediate device interrogation and contacting Medtronic Technical Support are crucial.
Device Malfunction During MRI
Should a LINQ II device malfunction become apparent during an MRI scan, the procedure must be immediately halted, and the patient’s condition stabilized. Potential malfunctions include electrical reset or inappropriate sensing, as outlined in the device’s procedural manual.
Prioritize patient assessment, monitoring vital signs, and providing necessary support. Following scan cessation, a thorough device interrogation is essential to determine the nature and extent of the malfunction.
Promptly contact Medtronic Technical Support for guidance and assistance in managing the situation. Document all events meticulously, including scan parameters, observed anomalies, and communication with support personnel, for comprehensive record-keeping and future analysis.
Post-MRI Device Interrogation
Following every MRI scan involving a LINQ II device, a comprehensive device interrogation is mandatory. This assessment verifies the device’s continued functionality and identifies any potential alterations caused by the MRI exposure.
Interrogation should encompass battery status, lead integrity, sensing thresholds, and stored events. Document all findings meticulously, comparing pre- and post-scan parameters to detect any discrepancies.
Any observed changes or abnormalities necessitate immediate evaluation and potential consultation with Medtronic Technical Support. Thorough documentation ensures a clear record of device performance and contributes to ongoing safety monitoring and data analysis.
Contacting Medtronic Technical Support
In instances of device malfunction post-MRI, or when interrogation reveals unexpected findings, prompt consultation with Medtronic Technical Support is crucial. Their expertise provides invaluable guidance for diagnosis and appropriate management strategies.
Medtronic’s support team can assist with interpreting device data, troubleshooting potential issues, and determining the necessity for device replacement or reprogramming.
Access to technical support is readily available through designated phone lines and online resources. Maintaining open communication with Medtronic ensures optimal patient care and adherence to the latest safety protocols regarding the LINQ II system.
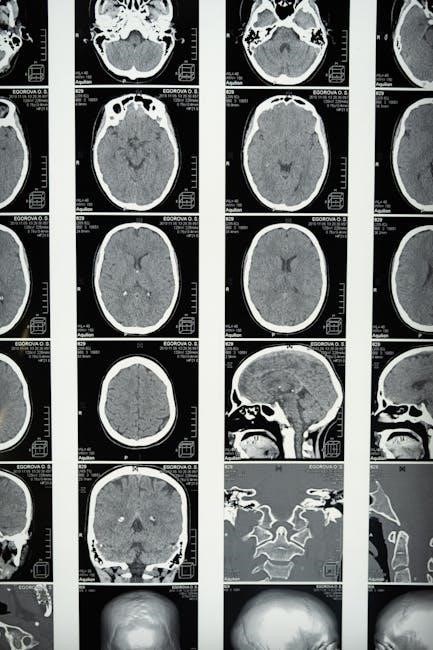
Documentation & Record Keeping
Detailed documentation of each MRI scan – including parameters, date, and any observed artifacts – is essential for maintaining a comprehensive device history.
MRI Scan Documentation Requirements
Comprehensive documentation is paramount when performing MRI scans on patients with the LINQ II Insertable Cardiac Monitor. Records must include the date and time of the scan, the specific MRI environment details (magnetic field strength, SAR limits), and the scan sequences utilized.
Crucially, document the implanted device model number (M301, M302, or M312) and any observed imaging artifacts. A detailed description of the artifact’s location and characteristics is vital for clinical interpretation.
Furthermore, record any post-scan device interrogations and their results, noting any changes in device function or battery status. Adherence to these requirements ensures a complete and traceable record for patient safety and future reference.
Device History & Scan Details
Maintaining a thorough device history is essential alongside detailed MRI scan information. This includes the LINQ II’s implantation date, model number (M301, M302, or M312), and any prior device interrogations or programming changes.
Scan details must encompass the MRI system manufacturer and model, the magnetic field strength employed, and the specific scan parameters utilized – including SAR values and scan sequences.
Documenting any observed artifacts, their location, and impact on image quality is crucial. This comprehensive record facilitates accurate clinical interpretation and supports ongoing safety monitoring of patients with the LINQ II ICM.
Adherence to Updated Information
The landscape of MRI safety and device compatibility is continually evolving. Therefore, strict adherence to the most current LINQ II MRI Technical Manual is paramount before each scan. Medtronic regularly updates this manual with new findings and refined recommendations.
Healthcare professionals must verify they are utilizing the latest version, ensuring patient eligibility assessment and scan protocol selection reflect the most up-to-date guidance.
Failure to do so could compromise patient safety and potentially lead to device malfunction or inaccurate image interpretation. Regularly check for updates and disseminate information within the clinical team.
Regulatory Information
The LINQ II ICM has received FDA clearance and expanded indications, including pediatric use. Medtronic provides comprehensive product safety information and complies with MRI safety standards.
FDA Clearance & Expanded Indications
The LINQ II Insertable Cardiac Monitor (ICM) has successfully obtained clearance from the Food and Drug Administration (FDA), signifying its safety and efficacy for intended use. Notably, Medtronic secured expanded indications in September 2022, broadening the device’s applicability to include pediatric patients aged two years and older.
This expansion allows for earlier detection of cardiac arrhythmias in a wider patient population. A key feature contributing to its clinical utility is MRI compatibility, enabling patients to undergo magnetic resonance imaging scans when medically necessary without device interference. The LINQ II’s design ensures it doesn’t impede daily activities, including water-based pursuits like showering, bathing, and swimming, enhancing patient quality of life.
Medtronic Product Overview & Safety Information
The LINQ II Insertable Cardiac Monitor (ICM), manufactured by Medtronic, is a small, implantable device designed for long-term cardiac rhythm monitoring. Crucially, patients with the LINQ II must avoid exposure to sources of diathermy, high radiation levels, electrosurgical cautery, external defibrillation, lithotripsy, therapeutic ultrasound, and radiofrequency ablation.
These exposures can potentially cause electrical reset of the device or trigger inappropriate sensing, as detailed in the Medical Procedure and EMI Warnings manual. MRI scans are permissible only within specified MR environments and adhering to the conditions outlined in the LINQ II MRI Technical Manual, ensuring patient safety and optimal device functionality.
Compliance with MRI Safety Standards
The LINQ II Insertable Cardiac Monitor’s MRI compatibility adheres to stringent safety standards, requiring careful adherence to the guidelines detailed within this technical manual. Prior to any MRI scan, verification of the patient’s eligibility and the specific MRI environment specifications is paramount.
Each scan necessitates confirming the device model number (M301, M302, or M312) and ensuring compliance with magnetic field strength and Specific Absorption Rate (SAR) limits. Continuous updates to this manual are crucial, as new information regarding MRI safety and device performance becomes available, guaranteeing optimal patient safety and data preservation.